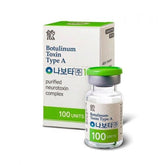
Our products are exclusively for use by medical professionals, and are not intended for personal use or self-treatment. The content on this website is for informational purposes only and should not be considered as medical advice or a substitute for professional consultation. Please consult with a qualified healthcare provider for treatment recommendations and safety information.